Redefining Rare Kidney Disase Treatments
Science that Transforms live
DETERA Therapeutics received SME status from EMA
DETERA Therapeutics @ LSX Lisbon March 2026
DETERA Therapeutics, winner of the Ipsen Golden Ticket 2025
DETERA Therapeutics received SME status from EMA DETERA Therapeutics @ LSX Lisbon March 2026 DETERA Therapeutics, winner of the Ipsen Golden Ticket 2025
DETERA Therapeutics @ BioLabs Hotel-Dieu, Paris
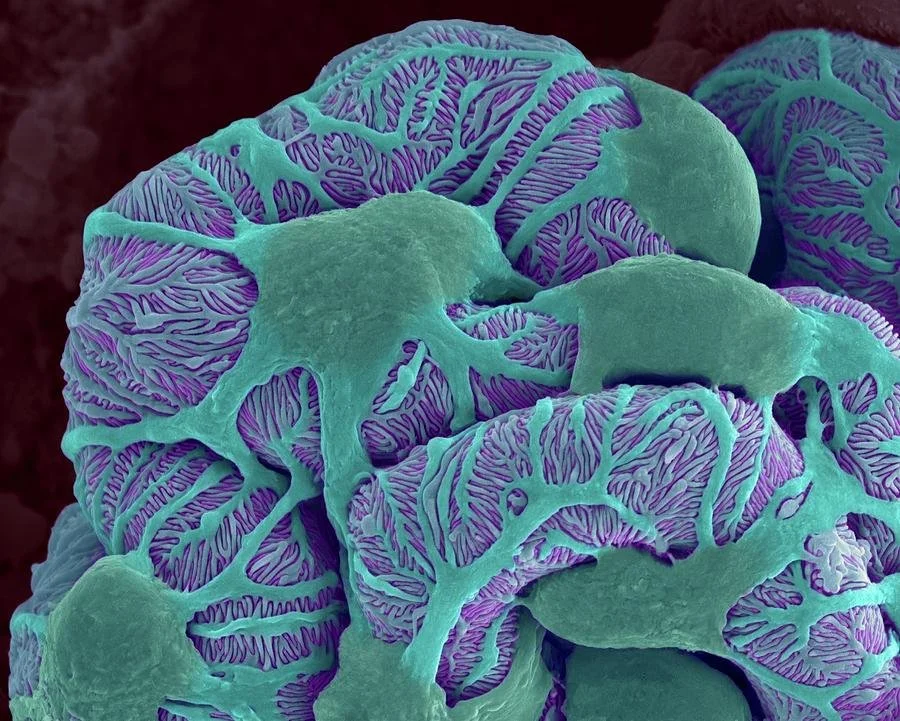
Pioneering the Future for Rare Neprhology with a Scalable Biotech Platform
We are an emerging player on the rare kidney diseases market for adults and children
OUR MISSION : Developing a portfolio of First-In-Class transformative therapeutic solutions
Our lead drug candidate DTR8 is developed for the treatment of Crescentic Glomerulonephritis (CGN), a severe condition with a high rate of therapeutic failure and associated costs.
DTR8 is a First-In-Class Disease-Modifying Drug for CGN. This is a first local, specific and immediate kidney therapy, with a compagnon test, allowing to stop the degradation of the kidney, preserve its function and decrease the number of patients under dialysis.
Redefining hope in rare kidney diseases
Meet DETERA Therapeutics @
Sections of the Website